Abstract:
The formation of scar tissue, or fibrosis, as gastric cancer disseminates throughout the peritoneum can be more lethal than the cancer itself and can interfere with chemotherapy. Researchers from Kanazawa University have now found that proinflammatory cytokine IL-17A from mast cells heavily influences the degree of fibrosis and causes structural changes in peritoneal cells. Preventing mast cells from releasing IL-17A may therefore be a promising treatment strategy for gastric cancer patients with peritoneal dissemination.
Kanazawa, Japan – Gastric cancer, one of the leading causes of cancer-associated mortality worldwide, is renowned for its ability to disseminate throughout the peritoneal cavity. As well as causing secondary tumors in other organs, metastatic gastric cancer cells trigger extensive stromal fibrosis, or the formation of scar tissue, that can be more deadly than the cancer itself—bowel obstruction and hydronephrosis and jaundice are all common side effects of gastric cancer-associated fibrosis. What’s more, the densely packed scar tissue can disturb chemotherapy drugs from reaching their target due to intra-tumoral high pressure.
Preventing fibrosis could therefore improve the prognosis for gastric cancer patients. The problem is, researchers have yet to discover what causes fibrosis, let alone how to prevent it.
But in a study published recently in Gastric Cancer, researchers from Kanazawa University found that an inflammatory protein produced by mast cells, IL-17A, triggers cellular changes in the peritoneum, leading to stromal fibrosis in gastric cancer patients.
Lead author Katsuya Gunjigake from Kanazawa University’s Division of Cancer Medicine explains why the researchers targeted IL-17A.
“Over-stimulation of the immune system by IL-17A plays a major role in chronic inflammatory diseases such as rheumatoid arthritis and multiple sclerosis. It has also been associated with increased tumor growth and dissemination in various forms of cancer. Interestingly though, while studies had shown that IL-17A causes fibrosis in both Crohn’s disease and lung disease, no one had investigated the link between tissue fibrosis and IL-17A in cancer.”
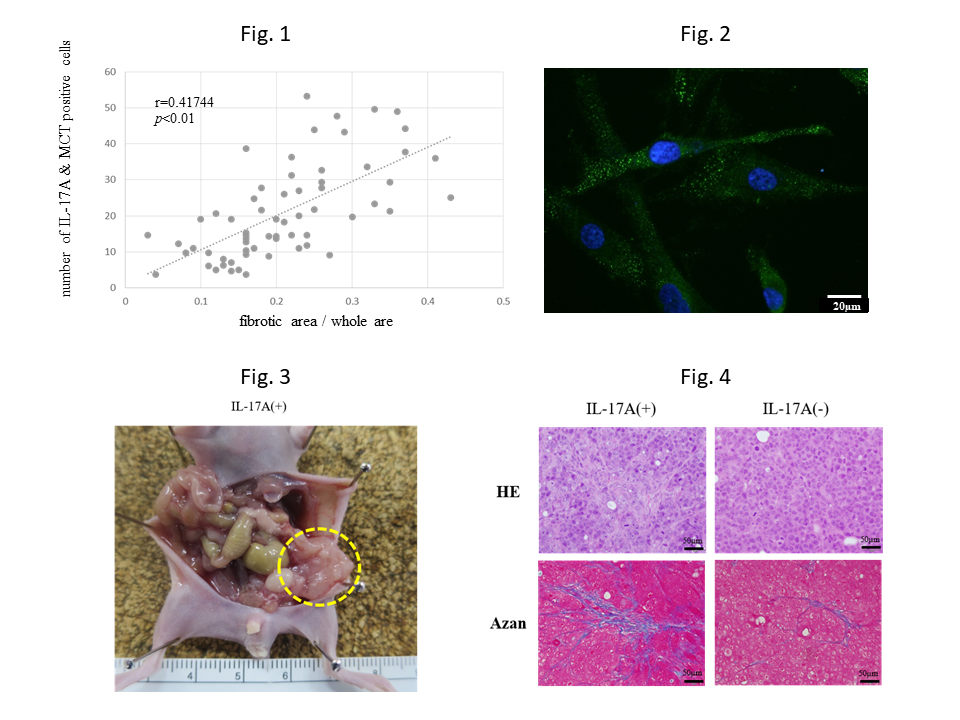
By studying cancerous tissue from 70 gastric cancer patients with peritoneal dissemination, the researchers discovered that the degree of fibrosis was governed by the amount of IL-17A, and that IL-17A was being produced by a subgroup of white blood cells called mast cells.
Says Gunjigake, “Mast cells are most commonly associated with anaphylaxis but are also involved in pathogen defense and immune tolerance, among other things. They contain small particles called granules that are filled with molecules such as histamine, serotonin, and IL-17A that are released into the extracellular environment in a process known as degranulation.”
The researchers then injected mice with human peritoneal cells and gastric cancer cells and examined the effects of IL-17A treatment, with interesting results.
“Not only did IL-17A increase tumor size and the degree of fibrosis, it also changed the structure of the peritoneal cells, enhancing their invasive and migratory capabilities,” explains responsible author Sachio Fushida.
“Given the obvious role of IL-17A in driving fibrosis, our results suggest that suppression of mast cell degranulation may be a promising treatment strategy for gastric cancer patients with peritoneal dissemination.”
Figure
Fig. 1 The number of IL-17A and MCT double positive cells correlated with the ratio of fibrotic area in the peritoneal tumors. This means IL-17A derived from mast cells may contribute tumor fibrosis. MCT: mast cell tryptase
Fig. 2 Expression of FAP was found in HPMCs treated with IL-17A using immunofluorescence staining. This means HPMCs were transformed into myofibroblast, so called CAF, by IL-17A. FAP: fibroblast activation protein as a marker of CAF. HPMCs: human peritoneal mesothelial cells. CAF: cancer associated fibroblast.
Fig. 3 Nude mouse was inoculated with human gastric cancer cell line MKN45-P cells intraperitoneally at day 0. Recombinant mouse IL-17A was administrated intraperitoneally at day 1, 3 and 7. Large and many peritoneal nodules were found at day 14.
Fig. 4 Fibrosis in the peritoneal nodules was recognized as blue area by azan staining. Fibrous area in the tumor treated with IL-17A was wider than that without IL-17A.
Article
Interleukin-17A derived from mast cells contributes to fibrosis in gastric cancer with peritoneal dissemination
Journal:Gastric Cancer
Authors:Katsuya Gunjigake, Jun Kinoshita, Takahisa Yamaguchi, Hiroto Saito, Daisuke Fujimori, Toshihide Horiike, Shinichi Harada, Hidehiro Tajima, Itasu Ninomiya, Tetsuo Ohta, Sachio Fushida
DOI: 10.1007/s10120-020-01092-2
Funder
This work was supported by JSPS KAKENHI Grant Number 16K10494.



 PAGE TOP
PAGE TOP