Abstract:
This study shows that, upon HCV infection, mRNA of selenoprotein P (SeP), a secretory protein produced primarily in the liver, binds to and inhibits the action of RIG-I, an antiviral protein, thus regulating innate immunity. RIG-I-mediated induction of interferon production is repressed by SeP mRNA, a host mRNA, affecting its antiviral activity. These results should lead to further elucidation of mechanisms underlying the association between metabolic disorders and virus-induced innate immunity dysfunction.
[Background]
Hepatitis C Virus (HCV)*1) is a hepatotropic virus, ~80 million chronic infections being confirmed worldwide. HCV infection leads to the development of chronic hepatitis, cirrhosis, and in some instances, hepatocellular carcinoma. The recent development of highly potent direct-acting antiviral drugs (DAAs)*2), targeting viral proteins, facilitates virus elimination in >90% of treated individuals. However, the mechanism of DAA failure in the remaining 10% is not understood.
The liver is a vital organ with functions in the metabolism of carbohydrates, proteins, and lipids, the clearance of toxins and pathogens, and the regulation of immune reactions. Previously, the present research group discovered selenoprotein P (SeP)*3), a liver-derived secretory protein; we reported that SeP contributes to the induction of insulin resistance and is involved in the pathogenesis of type 2 diabetes. Interestingly, patients with both chronic hepatitis C (CHC) and type 2 diabetes are reported to show a tendency to be unresponsive to DAAs. In the present study, the group investigated the pathogenic and clinical relationship of HCV infection and type 2 diabetes, focusing on SeP.
[Outline of the results]
The group at Kanazawa University investigated the effects of SeP on antiviral immune responses in the liver, using cultured cells, mice and clinical specimens.
It was found that HCV infection augmented SeP expression in cultured cells. By analyzing clinical specimens, it was found that serum SeP levels were higher in HCV-infected patients than healthy controls and that treatment with DAAs was less effective in patients with higher levels of SeP expression.
On the other hand, a remarkable repression of HCV replication was observed in the cultured cells with suppressed expression of SeP. These cells were also found to have an augmented production of interferon (IFN), which has antiviral activity. Humans are known to have a system to detect virus invasion into cells and to induce IFN production. It is reported that RIG-I*4) and MDA5 (melanoma differentiation-associated gene 5) recognize virus-derived RNA and induce IFN production. Although RIG-I and MDA5 have a highly homologous helicase domain*5), they recognize different RNAs depending upon RNA structure as well as base alignment (PAMP: pathogen-associated molecular pattern)*6). HCV is thought to be recognizable by both RIG-I and MDA5. By performing experiments using a RIG-I knockout cell line and a MDA5 knockout cell line, SeP was found to specifically regulate RIG-I pathway-dependent innate immune responses.
In more detailed experiments using purified SeP and synthetic RNA, repression of innate immunity by SeP was shown to be brought about not by the protein but by the SeP mRNA. Upon binding of a viral RNA to RIG-I, the ATPase activity of RIG-I is stimulated to induce a three-demensional structural change and oligomerization of RIG-I, which binds to mitochondrial antiviral signaling protein to activate IFN production. It was found that overexpression of SeP mRNA inhibited the ATPase activity important in the RIG-I activation steps: oligomerization of RIG-I and RIG-I localization to mitochondria. SeP mRNA, by binding to and interacting with the regulatory domain (RD) of RIG-I protein that specifically recognizes and captures viral RNA, inhibited the three-dimensional structural change necessary for RIG-I activation, and negatively regulated innate immunity.
Thus the present study has elucidated the regulation mechanism of innate immunity by SeP mRNA via RIG-I, one of the antiviral immune regulation mechanisms in the liver.
[Future prospects]
Since chronic hepatitis C and type 2 diabetes are high in morbidity worldwide, they must be treated as early as possible. In this study, we investigated the clinical relationship between HCV infection and type 2 diabetes, focusing on SeP, an inducer of insulin resistance. RIG-I protein was thought only to recognize RNAs of pathogens invading cells thus protecting against pathogens, but not to act on host RNAs. So far, very few host mRNAs have been reported acting like SeP mRNA, which functions as a ‘decoy’ to bind RIG-I thus repressing its functions. This information is important for understanding immune tolerance in the liver and autoimmune diseases. The study is expected to lead to further investigations of host RNAs regulating RIG-I and to the development of clinical application targeting such mRNAs, such as nucleic acid medicine.
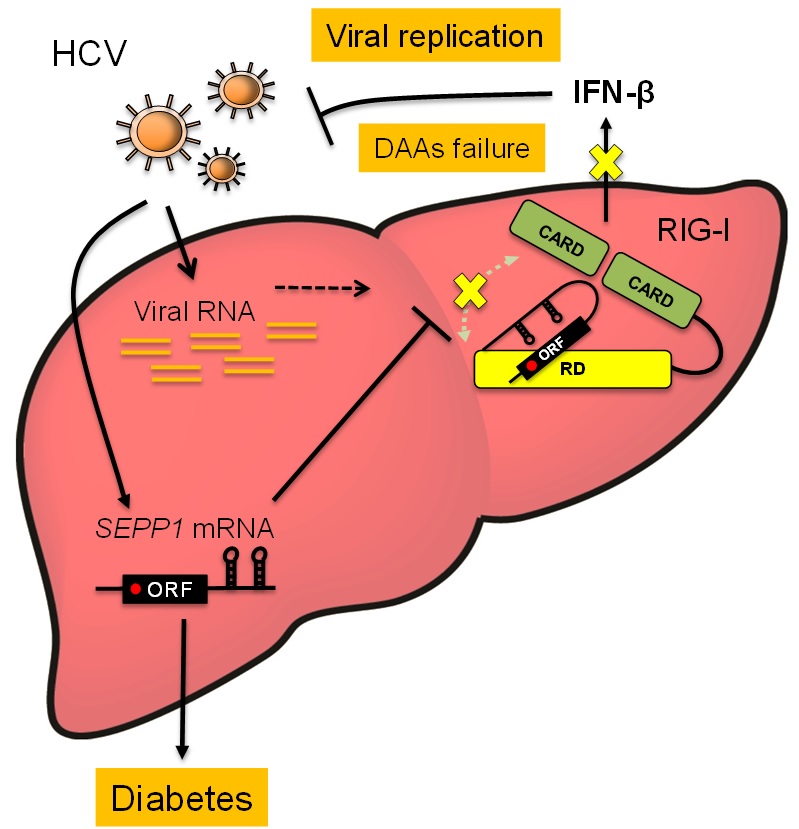
Figure. Regulation mechanism of innate immunity by SeP mRNA induced by HCV infection.
The liver infected by HCV augments selenoprotein P expression. Selenoprotein P gives rise to insulin resistance, accelerating pathogenesis of type-2 diabetes.
On the other hand, mRNA of selenoprotein P binds to the regulatory domain (RD) of RIG-I, inhibiting production of type I interferon (IFN-β) that has antiviral activity. As a result, viral replication is augmented which causes resistance against antiviral treatments.
[Glossary]
*1) Hepatitis C virus (HCV)
Hepatitis C virus (HCV), a member of the Hepacivirus C species, is a single-stranded RNA virus of the family Flaviviridae. The hepatitis C virus is the cause of hepatitis C.
*2) DAAs (direct-acting antiviral drugs)
DAAs are recently developed antiviral drugs that directly target viral proteins, having high antiviral efficacy.
*3) Selenoprotein P
Selenoprotein P is a protein that is responsible for transport of essential trace element Selenium (Se), produced primarily in the liver.
*4) RIG-I (Retinoic acid-inducible gene-I)
RIG-I is a protein that functions in the innate immune system. When a virus enters a cell, RIG-I recognizes virus-derived RNA and induces production of the type I interferon that exerts anti-virus activity.
*5) Helicase domain
Single-stranded RNA may fold to form a complicated structure and a part of the RNA may form a double-strand by self-binding. Helicases are class of enzymes that separate double-stranded parts of RNA and DNA into single-strands. The core part of a helicase with helicase activity is called the helicase domain.
*6) PAMP (pathogen-associated molecular pattern)
PAMP is a general name of pattern molecules specific to microbes (multicellular eukaryotes do not have such molecules). PAMP activates innate immunity. PAMP receptors are called pattern recognition receptors, including RLR (RIG-I-like receptor), TLR (Toll-like receptor) and NLR (NOD-like receptor).
Article
Induction of selenoprotein P mRNA during hepatitis C virus infection inhibits RIG-I-mediated antiviral immunity
Journal: Cell Host & Microbe
Authors: Kazuhisa MURAI, Masao HONDA, Takayoshi SHIRASAKI, Tetsuro SHIMAKAMI, Hitoshi OMURA, Hirofumi MISU, Yuki KITA, Yumie TAKESHITA, Kiyo-aki ISHII, Toshinari TAKAMURA, Takeshi URABE, Ryogo SHIMIZU, Hikari OKADA, Taro YAMASHITA, Yoshio SAKAI, and Shuichi KANEKO
DOI: 10.1016/j.chom.2019.02.015
Funder
Japan Agency for Medical Research and Development (AMED) JP18fk0210046, JP18fk0210012, JP18fk0210020, JP17fk0210201, JP18fk0210005, and JP18fk0310110; MEXT Grant-in-Aid for Scientific Research on Innovative Areas 17H06393.



 PAGE TOP
PAGE TOP