Researchers at Kanazawa University report in the Proceedings of the National Academy of Sciences of the United States of America (PNAS) high-speed atomic force microscopy experiments that show how ligands associated with stimulating and suppressing activation of the TRPV1 protein increase and decrease the molecule’s structural variations. The observations provide insights into how these heat- and chilli-sensing proteins function.
—————————————————————————————————————————————————————————————
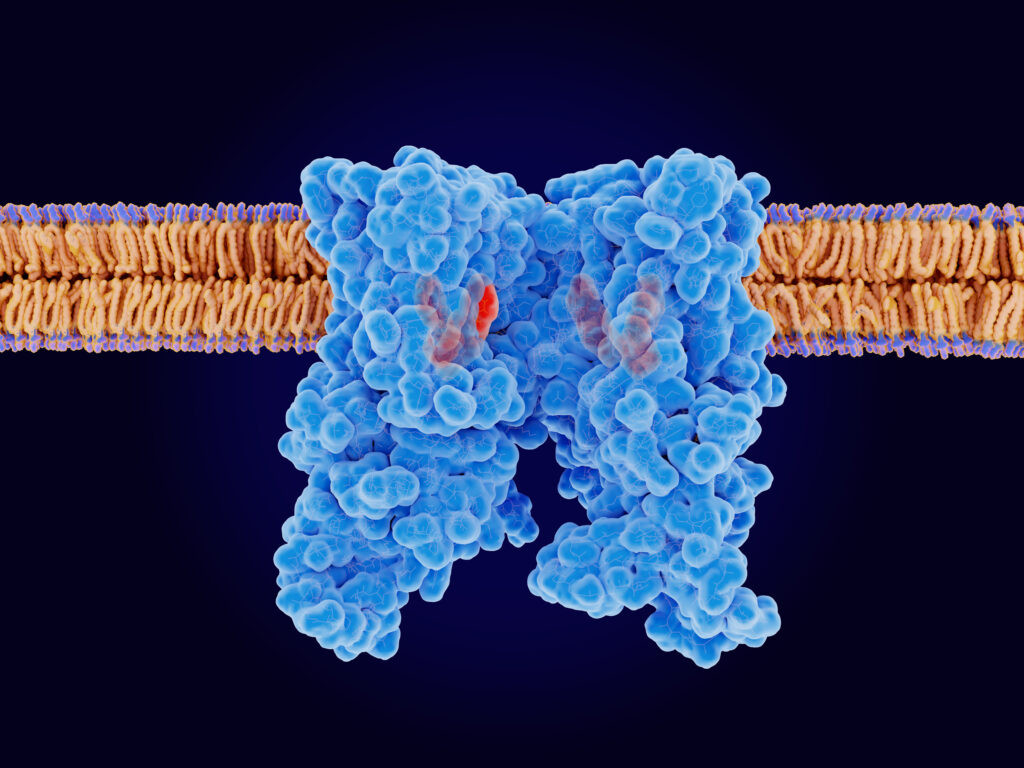
The capsaicin receptor TRPV1 with 4 ligand analogons bound. TRPV1 is an ion channel that senses heat, regulates the body temperature and contributes to pain sensation. Source: PDB entry 5is0. ©Juan Gärtner / stock.adobe.com
The skin senses heat – both from increased temperature and molecules like capsaicin in chillies – through the activation of protein receptors called Transient receptor potential vanilloid member 1 (TRPV1). However, the mechanisms behind the function of TRPV1 have not been clear. Now Ayumi Sumino at Kanazawa University in Japan and Motoyuki Hattori at the Fudan University in China and their colleagues provide important insights into this mechanism. Using high-speed atomic force microscopy to compare the protein with and without stimulating or suppressing molecules – ligands – bound to it, they obtain what they describe as “the first experimental evidence showing the correlation between molecular fluctuation and the gating state (ligand binding)”.
Once activated, the TRPV1 channel opens, allowing ions to permeate and signalling to the nervous system that a noxious stimulant is present. In 2011 researchers at the Howard Hughes Medical Institute in the US put forward a theoretical basis for the activation of the receptor derived from thermodynamics, a theoretical framework that has since been corroborated by experiment. The idea was that the molecule would respond to heat with a change in heat capacity, which is related to the fluctuations in the molecule’s conformation. Structures for the TRPV1 protein were known from previous cryo electron microscopy studies but these did not clarify how the fluctuations in protein conformation might change with stimulating or suppressing molecules, or even whether temperature and chilli sensing shared the same molecular mechanism.
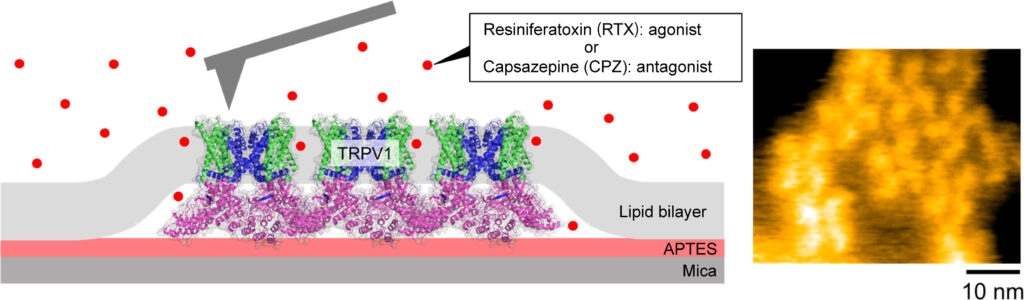
Atomic force microscopy (AFM) senses the topology of surfaces through the effect of distance on the forces on a nanosized tip positioned directly above the surface. The microscope was first invented in 1986 but gained a new lease of life through work at Kanazawa University that enabled it to capture topologies at high speed thereby providing a window into the dynamics of structures.
Sumino, Hattori and colleagues used high-speed AFM to image the TRPV1 receptor both in its unbound state and when bound to ligand molecules that either stimulate (agonist) or suppress (antagonist) the protein’s activity. They used the molecule resiniferatoxin, which is 1000 times hotter than capsaicin, as the agonist and for the antagonist they used capsazepine, which blocks the pain of capsaicin.
From the structures captured the researchers were able to observe fluctuations in the conformation of both the bound and unbound states of TRPV1. They found that resiniferatoxin increases conformational fluctuations, while capsazepine suppresses them.
Although the conformational fluctuations were very small – at around an Angstrom – the researchers highlight evidence in the literature of conformational changes at this scale being sufficient to affect the ion permeability of a channel. In their report of the work, the researchers conclude, “Overall, this study suggests the importance of structural fluctuation, which would be a key factor for the heat-sensing of TRPV1.”
Figure. Schematic illustration of HS-AFM imaging of the TRPV1 (left) and HS-AFM snapshot of TRPV1 channels (right).
Background
TRPV1
In 2021, David Julius shared the Nobel Prize for Physiology for his work in the 1990s, which led to the discovery of the role TRPV1 protein receptors play in sensing heat both from temperature and the capsaicin molecule in chillies. These proteins have been important for studies of pain because capsaicin provides such a well-controlled stimulus for heat pain.
High–speed atomic force microscope
Atomic force microscopy (AFM) “feels” the topography of surfaces by the increase and decrease of surface forces tugging on a nanoscale tip as changes in surface height change the distance between the tip and the surface. The tip is mounted on a cantilever so that the tiny changes in the force can be read out by the resulting deflection in the cantilever.
AFM can resolve structures with subnanometre scale resolution. It has particular advantages for biological studies because it does not require conducting substrates or a current, which are requirements for other primary microscopes with comparable resolutions, such as the scanning tunnelling microscope.
For a long time AFM was limited by the time it takes to capture an image of the surface, but this changed when Toshio Ando at Kanazawa University revealed how the tool could be upgraded to high-speed AFM using various modifications to the scanning, deflection detection and other electronic devices, as well as specifications for the cantilever. With high-speed AFM it became possible to capture dynamics at the nanoscale for the first time.
Reference
Ayumi Sumino, Yimeng Zhao, Daichi Mukai, Takashi Sumikama, Leonardo Puppulin, Motoyuki Hattori, Mikihiro Shibata. Antithetic effects of agonists and antagonists on the structural fluctuations of TRPV1 channel. Proceedings of the National Academy of Sciences. 120(20) e2301013120. May 8, 2023.
DOI:10.1073/pnas.2301013120
URL: https://doi.org/10.1073/pnas.2301013120



 PAGE TOP
PAGE TOP